Neonatal Clinical Trials
Session: Neonatal Clinical Trials 1
617 - Ethical Considerations for Using Alternative Methods of Consent in Neonatal Clinical Trials: Recommendations from a Modified Delphi Consensus Process
Saturday, April 25, 2026
3:30pm - 5:45pm ET
Publication Number: 2602.617
Stephanie Kraft, Geisinger College of Health Sciences, South Orange, NJ, United States; Abigail Brickler, Geisinger, Andreas, PA, United States; Abril Beretta, Seattle Children’s Childrens Hospital, Miami, FL, United States; Kaashif Ahmad, Pediatrix Medical Group, Houston, TX, United States; Justin Clapp, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, PA, United States; Neal W. Dickert, Emory University School of Medicine, Atlanta, GA, United States; Rebecca A.. Dorner, Sharp Mary Birch Hospital for Women & Newborns, San Diego, CA, United States; Elizabeth Field, Children's Mercy Hospitals and Clinics, Kansas City, MO, United States; Katherine Guttmann, Icahn School of Medicine at Mount Sinai, Larchmont, NY, United States; Anup Katheria, University of California, San Diego School of Medicine, San Diego, CA, United States; Andrea Kelsh, Columbia University, New York, NY, United States; Stephanie Kukora, Children's Mercy Bioethics Center, Kansas City, MO, United States; Diana Montoya-Williams, Childre's Hospital of Philadelphia, Philadelphia, PA, United States; Benjamin Mooso, University of California, San Diego School of Medicine, Sacramento, CA, United States; Muntasir Natour, Children's Hospital Los Angeles, Los Angeles, CA, United States; Laura Heck Ryan, Seattle Children's, Covington, WA, United States; Simran Sekhon, NA, KENT, WA, United States; Sandhya Subramanian, University of Washington School of Medicine, Seattle, WA, United States; Elliott Weiss, University of Washington School of Medicine, Seattle, WA, United States

Stephanie A. Kraft, JD (she/her/hers)
Associate Professor
Geisinger College of Health Sciences
South Orange, New Jersey, United States
Poster Presenting Author(s)
Background: Neonatal clinical trial recruitment can be challenging, particularly when enrollment decisions must be made quickly. Though alternative methods of consent may reduce enrollment bias and improve sample representativeness in this time-limited setting, the lack of prospective consent raises ethical concerns. Limited guidance exists in the US for balancing ethical tradeoffs when considering the use of alternative methods of consent in the neonatal intensive care unit (NICU).
Objective: To develop recommendations for the ethical use of alternative methods of consent in neonatal clinical trials.
Design/Methods: A multidisciplinary working group (n=16), including former NICU parents, neonatal clinical investigators, clinical research coordinators, research ethicists, equity scientists, and regulatory experts completed a modified Delphi process (Table 1). Initial statements were developed from relevant literature and a prior interview study with NICU parents about their views on alternative methods of consent. The working group reviewed each statement and rated their agreement on a scale of 1-9 ("very strongly disagree" to "very strongly agree"). Statements and group responses were discussed with the full working group, revised based on feedback, and re-rated by the group. Ratings were compared to previously determined consensus guidelines to produce a final list of consensus statements.
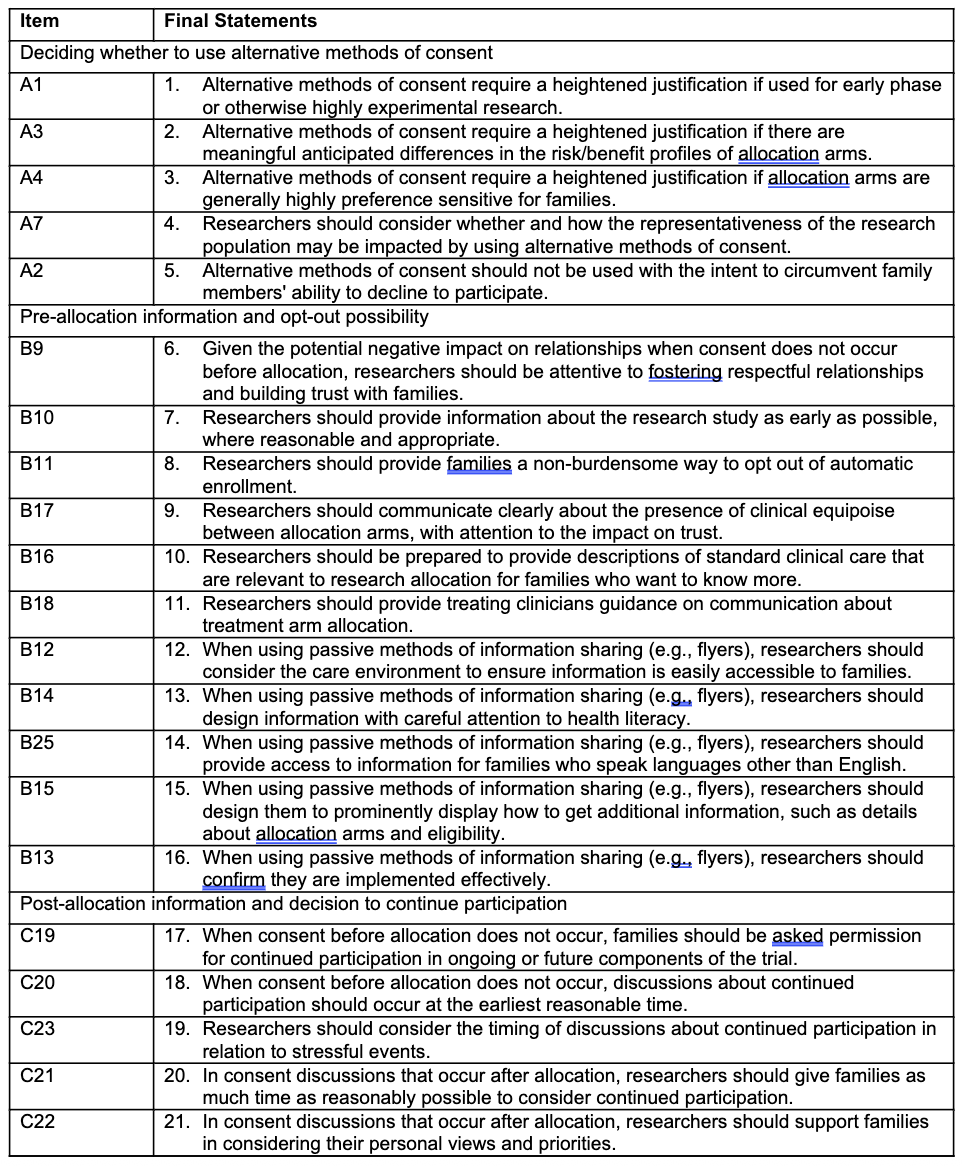
Results: Each statement was evaluated by 13-15 individuals in each round. From 24 initial statements (Table 2), 21 final statements reached consensus across 3 domains: (a) deciding whether to use alternative methods of consent, (b) pre-allocation information and opt-out possibility, and (c) post-allocation information and decision to continue participation (Table 3). Agreed-upon statements emphasized the importance of considering how alternative methods affect sample representativeness and, critically, relationships with families. They highlighted that communication about equipoise should be conducted in partnership with clinicians and prioritize trust. Finally, statements indicated that alternative methods may require a heightened justification in cases of early phase research, differential risk/benefit profiles, and high preference sensitivity; however, statements about heightened justification based on clinician preference or study outcomes did not reach consensus.
Conclusion(s): These consensus recommendations can guide neonatal researchers in the ethical use of alternative methods of consent. Further research on alternative consent methods in real-world settings is needed to evaluate downstream ethical implications.
Table 1. Steps for Modified Delphi Process
.png) FAM-CONNECT core team consisted of 4 individuals, two research coordinators, one neonatal bioethicist, and one expert in research ethics. FAM-CONNECT working group consisted of 16 individuals, including 3 former NICU parents and 13 others with expertise in neonatal clinical trials, clinical research coordination, research ethics, neonatal bioethics, regulatory affairs, and social anthropology.
FAM-CONNECT core team consisted of 4 individuals, two research coordinators, one neonatal bioethicist, and one expert in research ethics. FAM-CONNECT working group consisted of 16 individuals, including 3 former NICU parents and 13 others with expertise in neonatal clinical trials, clinical research coordination, research ethics, neonatal bioethics, regulatory affairs, and social anthropology. Table 2. Statements through Two Rounds of Surveys
.jpg)
Table 3. Final Consensus Statements